Services
Within our DMPK department, we house expertly managed in vitro & in vivo metabolite identification, and pre-formulation groups, led by seasoned scientists with a wealth of experience. Our dedicated teams are poised to navigate drug discovery projects by addressing scientific challenges, proposing pivotal modifications to chemical scaffolds, and steering projects towards their intended path.
By mitigating DMPK-related risks at an early stage, we strive to ensure a rapid and cost-effective journey toward successful drug development.
Comprehensive DMPK Services
At Jubilant, our DMPK technical and research services set the industry standard for excellence, tailored to meet the distinctive requirements of our valued clients. Our dedicated team offers unwavering support for integrated pre-clinical discovery programs and fixed-fee-for-services (FFS) projects, consistently delivering results that are both of exceptional quality and reproducibility, all within rapid turnaround times. We prioritize close collaboration with our clients to comprehensively understand their requirements and expectations with regard to time and cost, ensuring a transparent and tailored approach to every project.
Whether you need exploratory ADME screening or detailed pharmacokinetic studies, Jubilant offers a seamless, end-to-end DMPK solution.
Key Differentiators
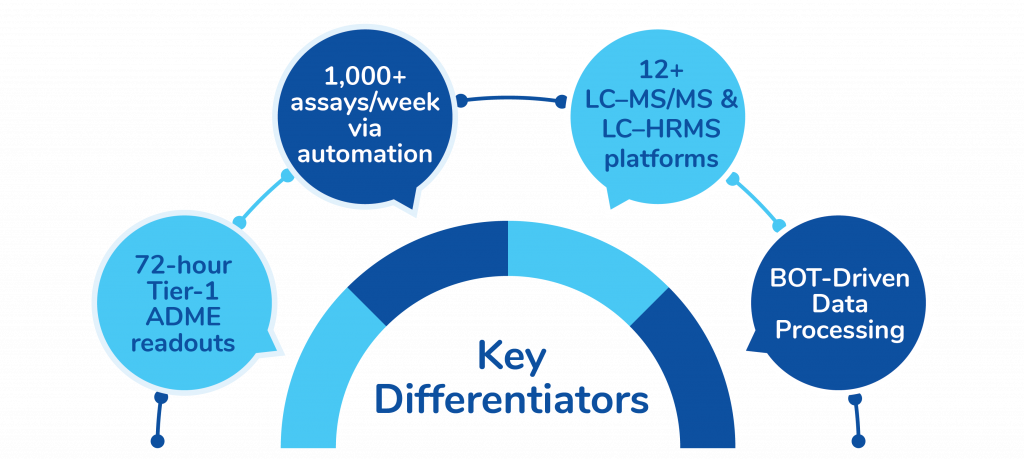
In vitro DMPK Services
Our team specializes in providing end-to-end in vitro ADME services, covering all key areas—absorption, distribution, metabolism, and excretion. With a wealth of experience, we provide expert guidance throughout the drug discovery journey, spanning from the initial hit identification phase to lead optimization, and ultimately culminating in the identification of clinical candidates.
Our in-depth in vitro ADME services are designed to provide valuable insights and data that help address potential issues and enhance the efficiency of your drug development endeavors.

Key areas of expertise within our In Vitro ADME services include:
Physiochemical Studies:
In Vitro Metabolism Services:
In Vitro Permeability and Transporter Studies:
Our in vitro DMPK scientists deliver high-throughput, reproducible data that help streamline candidate prioritization and predict human pharmacokinetics early in the pipeline.
In vivo DMPK and Pharmacokinetic Services
Our comprehensive in vivo services encompass a wide spectrum of studies to evaluate the pharmacokinetics of compounds. We conduct pharmacokinetic studies using various administration routes, including oral, intraperitoneal, intravenous, subcutaneous, and more, in a range of species, including mice, rats, rabbits, minipigs, and dogs.
Our in vivo services extend to:
Additionally, we offer specialized studies, including:

Our in vivo pharmacokinetic experts design studies that ensure translational accuracy between preclinical models and human systems, using precise bioanalytical quantification methods to deliver actionable PK insights.
Novel Therapeutic Modalities

Why Choose Jubilant Biosys for DMPK Services
Choosing Jubilant means partnering with a trusted DMPK CRO that combines decades of experience with integrated drug discovery expertise. Our collaborative approach ensures each study is aligned with project goals, timelines, and regulatory expectations.
Why global pharma and biotech companies trust Jubilant Biosys:
- 20+ years of expertise in discovery and preclinical DMPK
- Integrated collaboration with medicinal chemistry, bioanalysis, and toxicology teams
- State-of-the-art analytical platforms (LC-MS/MS, high-throughput automation)
- Rapid data delivery and transparent communication
- Proven experience across small molecules and biologics
By partnering with Jubilant, you gain a strategic DMPK partner capable of delivering precise, decision-enabling data that accelerates discovery timelines.
Scientific Expertise and Publication
Our DMPK scientists are active contributors to the scientific community, with numerous publications in high-impact, peer-reviewed journals and poster presentations at international conferences. This ongoing research reinforces Jubilant’s position as a leader in drug metabolism and pharmacokinetic studies, ensuring that our methodologies remain at the forefront of innovation.
Connect with Our DMPK Experts
Ready to advance your discovery program with high-quality DMPK services? Contact our team to discuss your study requirements or request a customized proposal.
Partner with Jubilant Biosys for accurate, efficient, and integrated in vitro and in vivo DMPK services that help you make confident, data-driven decisions in your drug discovery journey.
